Instrumentation for Wavelength Calibration
Authors: Xiaohua Ye, Megan Dube
Spectrometers and other wavelength-sensitive instruments will naturally drift due to environmental effects and influences over time. This issue is common for these types of instruments. It can be addressed by regular wavelength calibration, which helps to resume accurate and reliable spectroscopic results. Wavelength calibration can be achieved by observing the spectrum of emission lines of known wavelengths from a designated instrument, including gas-discharge lamps, laser lines, tunable light sources, and more. In this note, a discussion and comparison of the instruments used in wavelength calibration is presented to better understand the performance and limitation of different light sources for the application of wavelength calibration.
Typical Equipment
- Spectral lamps
- Laser lines
- Tunable light sources
- Liquid filters/Glass filters
- Light from sun
Typical Equipment for Wavelength Calibration
1. Spectral Lamps
Spectral lamps used for wavelength calibration include pencil style calibration lamps, larger bulb spectral lamps, and hollow cathode lamps.
1.1 Pencil Style Calibration Lamps
Pencil style calibration lamps (also called pen-ray lamps) are versatile and feature a compact layout that can easily be integrated into an optical system for wavelength calibration. Spectral lines from typical rare gases and metal vapor include the elements of Ar, Kr, Xe, Ne, Hg, and the combination of Hg with other elements. Because of this, the UV-VIS-NIR spectrum ranging from 230 nm to 2000 nm can be covered. Figures 1 and 2 show the typical line output in the VIS range of 600-700 nm and 410-740 nm for Krypton and Argon lamps, respectively. Tables I and II list the strong lines of Xenon at 800-860 nm and Mercury (Hg) at 200-300 nm. These lamps are popular options for wavelength calibration applications due to their versatility and lower cost compared to other solutions.
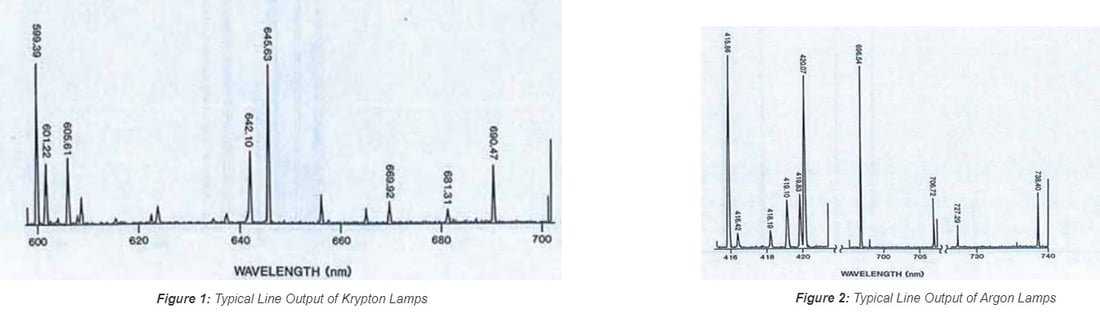
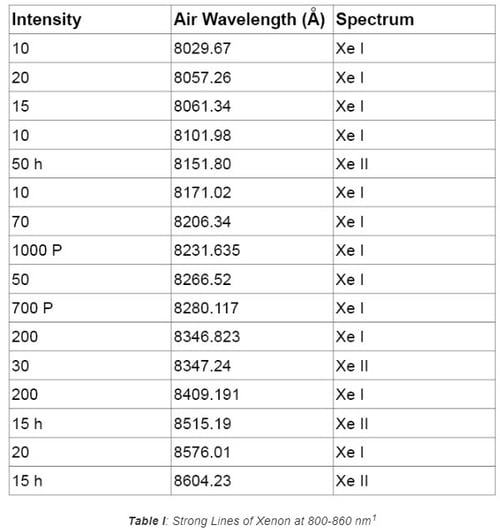
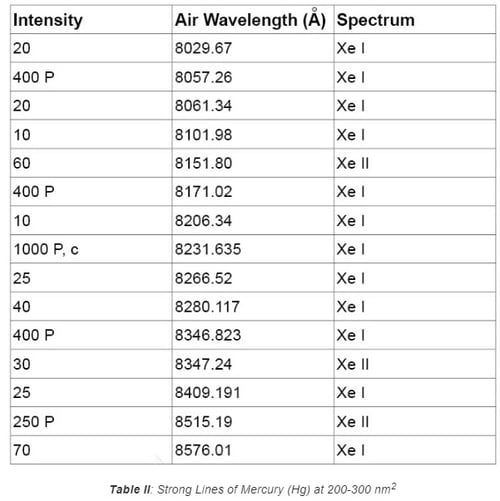
1.2 Hollow Cathode Lamps
Hollow cathode lamps are a type of discharge lamp that consist of the following: a cathode made from an atomic element designed for desired spectral output, an anode, and an inert gas contained in the sealing glass tube of the lamp. In operation, the spectral lines are generated from the filler gas atoms that are ionized when a specific designed voltage is applied and the sputtered atoms from the cathode which are excited in the plasma output. Typical elements used as the electrodes include As, Cs, Sb, Cd, Sn, Si, Ni, Te, Hg, Ba, and Rb. The inert gas that is generally used is either Ar or Ne.
Spectral output for hollow cathode lamps presents a low intensity, no continuum, and a large number of weak spectral lines. For specific applications, these lamps can deliver spectral lines for virtually every 5 nm interval in the UV and VIS spectral ranges, which enables accurate wavelength calibration if the instrument under test is sensitive enough.
1.3 Large bulb Spectral Lamps
Compared to pen-ray lamps, large bulb spectral lamps are generally more intense in terms of light output. Large bulb spectral lamps typically utilize Zn, Hg, Cd, Ar, Xe, Kr, Rb, Na, He and the combination of Hg with other elements as lighting medias. The spectral range of these lamps can cover a UV-VIS-NIR region of 210 nm to 2300 nm.
2. Laser Lines
Laser lines can also be used for wavelength calibration. For instance, He-Ne emissions provide laser lines at 543.37 nm, 593.93 nm, 611.80 nm, 632.82 nm, and an NIR line at 1523 nm. An argon fluoride laser can generate a UV line at 193 nm, while He-Cd lasers are capable of spectral lines at 325 nm and 442 nm. In Table III, typical laser lines from different lasers ranging from UV to IR are listed. From a cost perspective, using laser lines for wavelength calibration is typically more expensive than using gas lamps. A gas lamp also contains more lines compared to a laser. However, the intensity of laser lines is much higher than the emission lines from gas lamps.
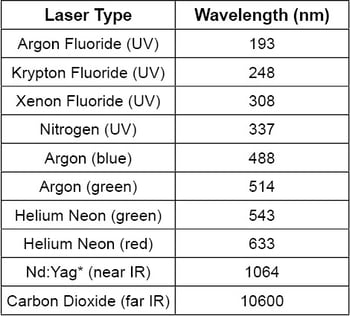
Table III: Commonly Used Lasers and Their Emission
3. Tunable Light Sources with Monochromatic Output
Tunable light sources built with broadband light sources, such as quartz tungsten halogen lamps or Xe lamps and monochromators, can produce a monochromatic and tunable light output. This output can be coupled with an optical fiber for more convenient deployment. Once calibrated, the tunable light source is capable of providing an infinite number of spectral lines delivered by the monochromator. The bandwidth of the monochromatic output, commonly expressed as “full width at half maximum” (FWHM), is determined by the slit width of the entrance and exit slits of the monochromator. Typical FWHM is greater than 0.1 nm, which is wider than the width of the atomic lines. Wavelength scale calibration determines the accuracy of the spectral lines. The contrast ratio or purity of the spectral output is determined by the straylight rejection performance of the monochromator. The spectral range for the system is limited by the spectral output for the light source and tunable range of the monochromator, typically covering 200 nm to the NIR region up to 3000 nm.
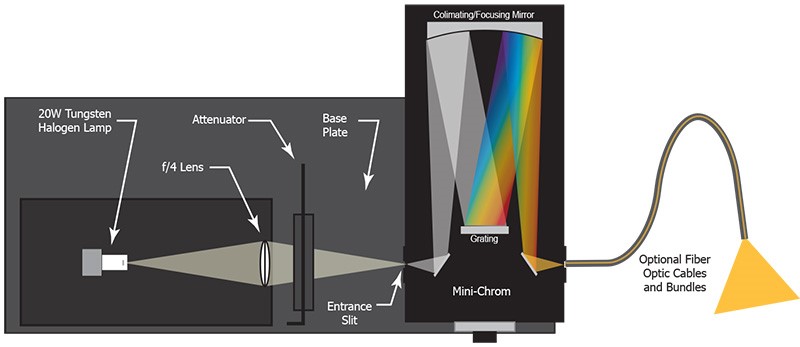
Figure 3: Layout for a Typical Tunable Light Source
4. Liquid Filters and Glass Filters
Liquid filters and glass filters reduce the light beam of the spectrophotometer to a greater extent at certain wavelengths with narrow, well-defined peaks for wavelength accuracy measurement. The reference material for liquid filters could be Cerium dissolved in perchloric acid for a spectral range of 200-260 nm, or a solution of Holmium oxide dissolved in perchloric acid to cover the 240-650 nm range.3 Figure 4 presents a typical spectrum for a Didymium liquid filter which utilizes praseodymium and neodymium dissolved in perchloric acid for checking the wavelength accuracy at 320-870 nm. Glass filters with similar reference material as liquid filters exhibit similar spectral output while having a slight difference in the intensity of some peaks. Compared to liquid filters, the main advantage of using glass filters is that they are more robust.
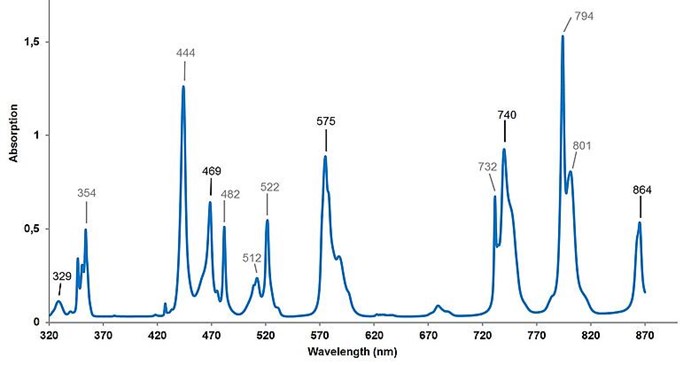
Figure 4: Typical Spectrum for Didymium Liquid Filter
5. Light from the Sun
The Sun emits radiation from x-rays to radio waves. Its spectrum consists of numerous absorption lines which are well characterized, identified, and accurately measured from space. The solar spectrum could be an alternative method of wavelength calibration. In practice, sunlight is collected and measured by the instrument under test with the aid of software. This software is used to apply shift and stretch corrections to the measured spectrum until the whole spectral structures match the tabulated lines of a solar reference spectrum. The acquired result presents the inaccuracy of the wavelength scale that needs to be corrected within the spectral range under investigation. The spectral range for wavelength calibration using sunlight typically applies from the UV to VIS range. This method does have its limitations, however, with such factors as weather and timing coming into play.
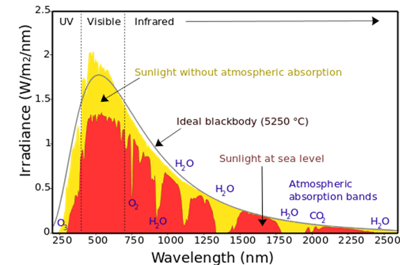
Figure 5: Solar Spectrum and Atmospheric Absorbing Gases from 240 nm to 2.5 µm Wavelengths
Summary
A summary of popular instrumentation and specifications for the application of wavelength calibration are shown in Table IV. In general, spectral lamps are the most common solutions for this application while other methods can be helpful to supplement spectral lamps in terms of spectral range, accuracy, and cost.
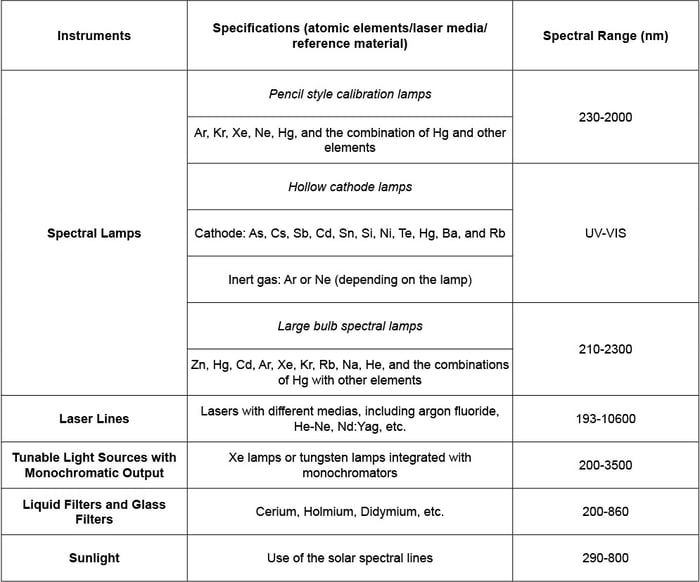
Table IV: Summary of Popular Instrumentation and Specifications for Wavelength Calibration
Sources
- Basic Atomic Spectroscopic Data. Strong lines of Xenon (XE). (n.d.). Retrieved April 3, 2023, from https://physics.nist.gov/PhysRefData/Handbook/Tables/xenontable2.htm
- Basic Atomic Spectroscopic Data. Strong lines of mercury (hg). (n.d.). Retrieved April 3, 2023, from https://physics.nist.gov/PhysRefData/Handbook/Tables/mercurytable2.htm
- Didymium liquid filter (320 - 870 nm) for checking the wavelength accuracy. Didymium Liquid Filter. (n.d.). Retrieved April 3, 2023, from https://www.hellma.com/en/laboratory-supplies/uvvis-reference-materials/checking-wavelength-accuracy/didymium-liquid-filter/


